Carbonic acid carbon dioxide formula12/5/2023  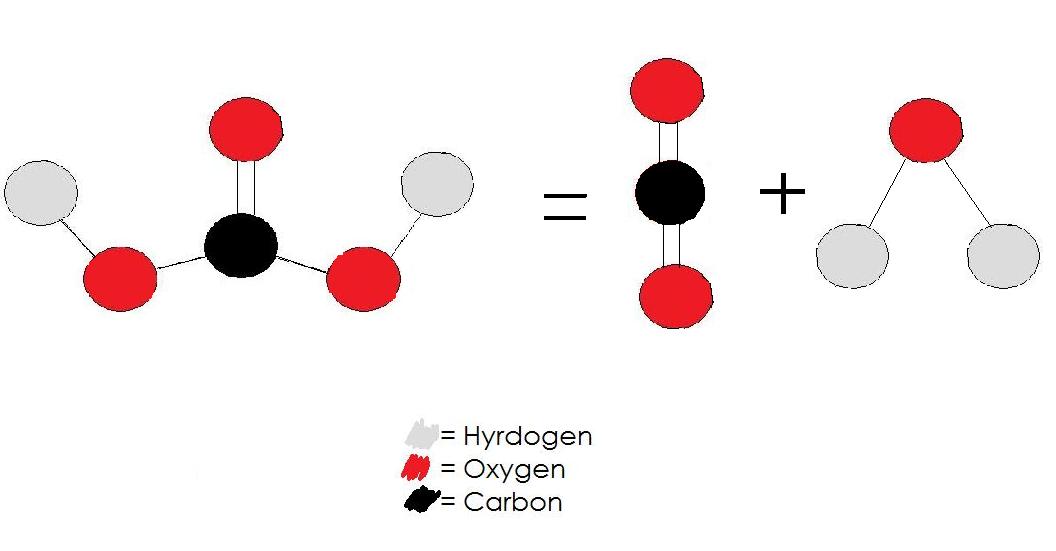
Experimental Ionization Potent: 13.77 Ev NIOSH FF6400000.Experimental Boiling Point: -78 ☌ (Sublimes) OU Chemical Safety Data (No longer updated) More details.78.48 ☌ (Sublimes) Kaye & Laby (No longer updated) Experimental Melting Point: -56.5 ☌ Jean-Claude Bradley Open Melting Point Dataset 22473.Experimental Physico-chemical Properties.The carbonic acid compound may have different names depending on the various different situations of industrial applications. Other names (synonyms) or registry numbers of carbonic acid.The InChIKey may allow easier web searches for carbonic acid, but it needs to be linked to the full InChI to get back to the original structure of the carbonic acid since the full standard InChI cannot be reconstructed from the InChIKey. The condensed, 27 character standard InChIKey (hashed version of the full standard InChI) of carbonic acid is: It can provide a standard way to encode the molecular information of carbonic acid to facilitate the search for the compound information in databases and on the web. The full standard InChI of carbonic acid is: An alternative textual expression including the structural information is InChI. The molecular chemical formulas lack structural information. InChI (IUPAC International Chemical Identifier) information of carbonic acid.The molecular weight of carbonic acid is available in molecular weight page of carbonic acid, which is calculated as the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element specified in the chemical formula of carbonic acid. The chemical structure image of carbonic acid is available in chemical structure page of carbonic acid, which specifies the molecular geometry, i.e., the spatial arrangement of atoms in the chemical formula of carbonic acid and the chemical bonds that hold the atoms together. The carbonic acid structure data file can be imported to most of the cheminformatics software for further analysis and visualization. The structure data file (SDF/MOL File) of carbonic acid is available for download in the SDF page of carbonic acid providing the information about the atoms, bonds, connectivity and coordinates of carbonic acid, which is not completely available in the chemical formula representation. Structure Data File (SDF/MOL File) of carbonic acid. 
QSPR ModelingĪdditional Information for Identifying Carbonic acid Molecule.Quantum Tools for IR Spectra Interpretationįaster Quantum Calculations with Pre-Computed ResultsĬhemical Big Data for Artificial Intelligence (AI) Developments Thermophysical Property Datafile (IK-Cape File) for Process Simulators, e.g., Aspen Plusįree Radicals Thermodynamic Data for Oxidation, Combustion, and Thermal Cracking Kinetics Thus, each side of the chemical equation must represent the same quantity of any particular element based on the chemical formula.Įnter another compound to search for formula: The law of conservation of mass dictates that the quantity of each element given in the chemical formula does not change in a chemical reaction. The above chemical formula is the basis of stoichiometry in chemical equations, i.e., the calculation of relative quantities of reactants and products in chemical reactions. The chemical formula of carbonic acid shown above is based on the molecular formula indicating the numbers of each type of atom in a molecule without structural information, which is different from the empirical formula which provides the numerical proportions of atoms of each type. A chemical formula of carbonic acid can therefore be written as: There are 2 Hydrogen atom(s), 1 Carbon atom(s), and 3 Oxygen atom(s). The carbonic acid molecule contains a total of 6 atom(s). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
